Molecular fMRI
Sensors capable of imaging a specific molecular processes across the living brain, provide spatially resolved mechanistic information while accepting more of the complexity of whole living organisms. The added structural information and spatiotemporal dynamics, in turn takes advantage of our growing capacity to computationally analyze high dimensional datasets. Our ability to interpret the interplay of biological mechanisms as more than static pathways, but as dynamic molecular circuits, may be the key to understanding, and treating, the complex nature of neurodevelopmental and degenerative diseases.
ManICS
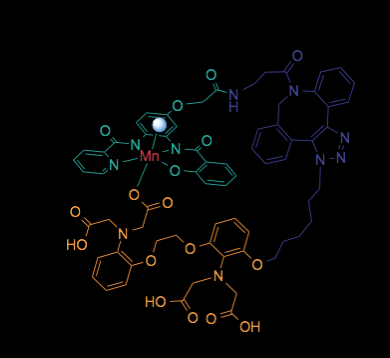
Calcium ions are essential to signal transduction in virtually all cells, where they help coordinate biological processes ranging from embryonic development to neural function in the brain. Although optical probes for intracellular calcium imaging have been in use for decades, the development of probes for noninvasive monitoring of intracellular calcium signaling in deep tissue and intact organisms remains an important challenge. To address this problem, we synthesized and characterized a manganese-based paramagnetic contrast agent, ManICS1-AM, that can be internalized and trapped in cells, with the aim of allowing intracellular calcium levels to be monitored by magnetic resonance imaging (MRI). Cells loaded with ManICS1-AM show changes in MRI contrast when stimulated with pharmacological agents or optogenetic tools. These responses directly parallel signals obtained using fluorescent calcium indicators, validating ManICS1-AM as an MRI-detectable analog of powerful optical probes.
Barandov A*, Bartelle BB*, Williamson CG, Loucks E, Jasanoff AP. Sensing intracellular calcium ions using a manganese-based MRI contrast agent. Nat Commun. 2019 Feb 22;10(1):897
MaCaReNa

Magnetic calcium-responsive nanoparticles (MaCaReNas) that can be detected by magnetic resonance imaging (MRI). MaCaReNas respond within seconds to [Ca2+] changes in the 0.1-1.0 mM range, suitable for monitoring extracellular calcium signaling processes in the brain. We show that the probes permit repeated detection of brain activation in response to diverse stimuli in vivo. MaCaReNas thus provide a tool for calcium activity mapping in deep tissue and offer a precedent for development of further nanoparticle-based sensors for dynamic molecular imaging with MRI.
Okada S*, Bartelle BB*, Li N, Breton-Provencher V, Lee J, Rodriguez E, Melican J, Bricault S, Sur M, Jasanoff AP. Calcium-dependent molecular fMRI using a magnetic nanosensor.Nature Nanotechnology 2018 Jun;13(6):473-477
Biotag
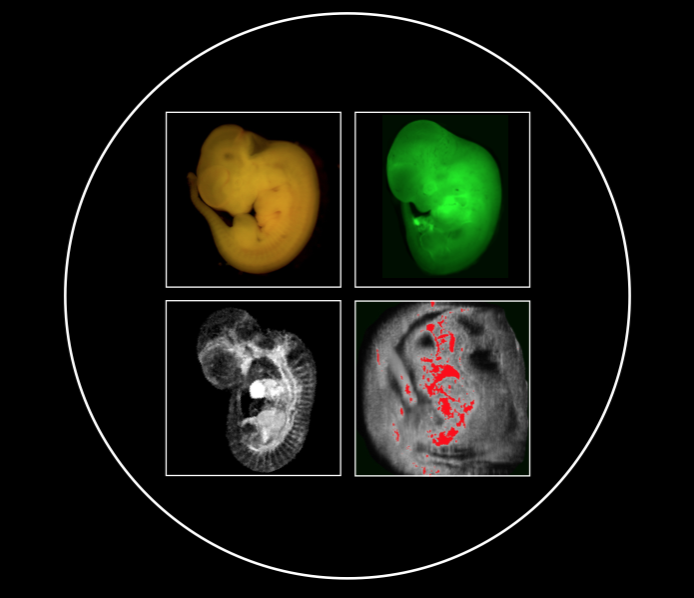
Biotag is a genetically expressible tag that can function as a multimodal reporter when coupled with an avidin coupled probe. The first application of Biotag is the Ts-Biotag mouse, which Biotag when the gene Tie2 is activated in vascular endothelial cells and in myeloid derived cells such as microglia. Activation of Tie2 occurs in a rapid response to wound signaling like peroxides and the cytokine Ang1, effectively making the Ts-Biotag system a signaling reporter, or sensor, for these molecules. We are using our Ts-Biotag mouse to dissect the neuroinflammatory signaling axis between microglia, astrocytes and the neural stem cell niched in response to neural injury. The goal of this work is to determine the pathways and contexts in which neuroinflammation can have negative or beneficial effects on wound healing and neural regeneration.
Bartelle BB, Berríos-Otero CA, Rodriguez JJ, Friedland AE, Aristizábal O, Turnbull DH. Novel genetic approach for in vivo vascular imaging in mice. Circulation Research. 2012 Mar 30;110(7):938-47. PubMed PMID: 22374133; PubMed Central PMCID: PMC3319022.
MR Reporters
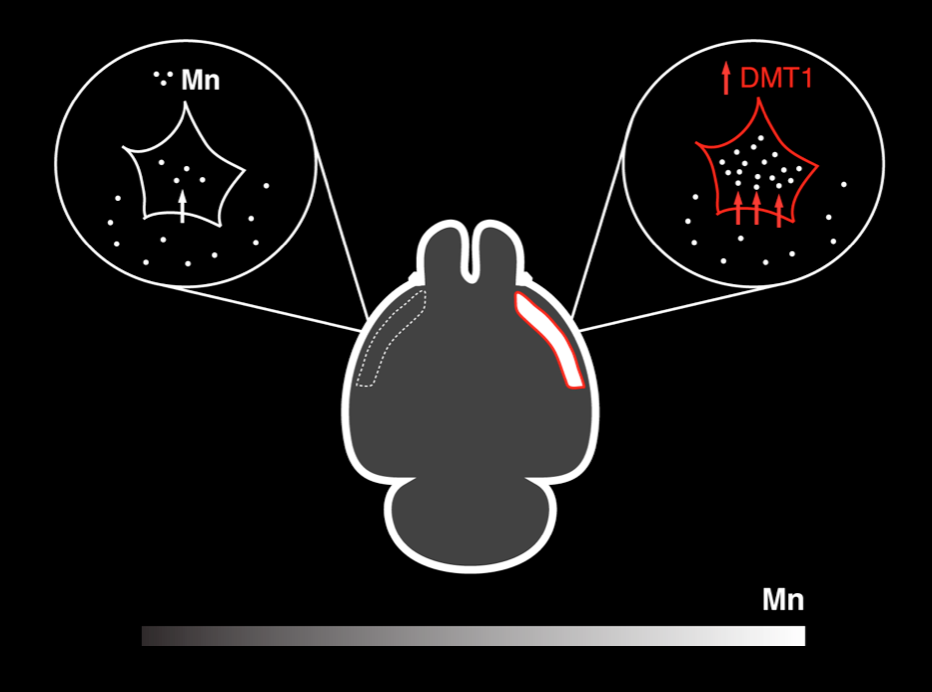
There remain very few robust genetic reporters for MRI with Biotag as the only current technology to be used in an actual research application. The lack of MRI reporter genes remains the greatest technical barrier for Molecular fMRI. We continue to explore biological modes of MRI contrast and dynamic effector mechanisms using protein engineering and synthetic biology along several lines. Manganese is a biologically endogenous MRI contrast agent with mechanisms that we have engineered to accumulate and modulate its MR properties dynamically in a living cell.
Bartelle BB, Mana MD, Suero-Abreu GA, Rodriguez JJ, Turnbull DH. Engineering an effective Mn-binding MRI reporter protein by subcellular targeting. Magnetic Resonance in Medicine. 2015 Dec;74(6):1750-7. Epub 2014 Dec 17. PubMed PMID: 25522343; PubMed Central PMCID: PMC4470876.
Bartelle BB, Szulc KU, Suero-Abreu GA, Rodriguez JJ, Turnbull DH. Divalent metal transporter, DMT1: a novel MRI reporter protein. Magnetic Resonance in Medicine. 2013 Sep;70(3):842-50 Epub 2012 Oct 12. PubMed PMID: 23065715; PubMed Central PMCID: PMC3587026.